 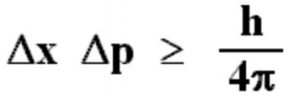
So we can never measure both factors simultaneously. So, in any case, either position or momentum/velocity can be measured accurately. Similarly, in the measurement of momentum, the error occurs in the position. Hence, in the case when a position is kept exact, the error occurs in the measurement of momentum or velocity. It means while fixing the position, the particle’s momentum would surely have changed from the original value. At the time of identification, a collision of a powerful light source increases the momentum of the electron and makes it move away from the initial position. For that, we must require an illuminating source (as a source of light to identify and measure the electron’s position). So electrons are not visible by naked eyes. Whereas in the case of microscopic particles, it would not be that easy to determine both the position and momentum simultaneously.Īs an atom consists of electrons, electrons have an atomic mass of 9.91×10 ‐ ³¹ kg. For example, The speed and location of a moving automobile can be determined simultaneously at once with very low or a minimum error. Momentum and position of macroscopic nature can be easily and simultaneously identified with accuracy. 
The uncertainty principle is only applicable to dual-natured microscopic particles, not macroscopic particles.Įxample of Heisenberg’s uncertainty principleĪs we know, electromagnetic waves and matter particles exhibit dual nature, i.e. If the position of an electron is subjected to be measured accurately to its size (10 -10 ), then the error(in the measurement of velocity) would be nearly equal to 1000 kilometres. The accurate measurement of position/momentum indicates a large error in the measurement of other quantities.Īpplication of Heisenberg’s principle on an electron in an orbit of the atom. ∆v is the measurement error of velocity assuming that the mass remains constant during the whole experiment, 
Heisenberg’s uncertainty formula can also be written as Heisenberg’s Uncertainty Principle FormulaĪssuming that ∆x is the error in the position measurement and ∆p is the measurement error of momentum then, This principle explains that simultaneous measurement of position and velocity of matter waves will always have an error. It states that the velocity and position of an object can not be measured precisely or simultaneously, and that’s because exact velocity and exact position have no meaning in nature. Heisenberg’s uncertainty principle, also called the indeterminacy principle, was given by German physicist Werner Heisenberg in 1927. Heisenberg’s Uncertainty Principle: Definition Whereas a particle wave having a well-defined wave is spread out, the particle associated with such a wave will have precise velocity and may be present anywhere. The particle associated with such a wave will have a definite position but no definite velocity. This is the reason a strictly localized wave has an intermediate wavelength. As a result, the momentum of the particle is determined. The more intense the undulations, the more ill-defined the wavelength becomes. 
The particle is observed at the place where the wave undulations are greatest. The uncertainty principle evolved from the wave-particle duality, i.e. This principle is contrary to Newtonian physics. Theoretically, both the velocity and position of an object can not be measured simultaneously, which is what this principle implies. This principle has been present in physics for a long time, making its importance too high. The uncertainty principle implies that it is in general not possible to predict the value of a quantity with arbitrary certainty, even if all initial conditions are specified.In quantum mechanics, Heisenberg’s uncertainty principle is the most important principle, which states that it is very difficult or nearly impossible to measure more than one quantum variable (as it is defined by its wave function) simultaneously. Such variable pairs are known as complementary variables or canonically conjugate variables and, depending on interpretation, the uncertainty principle limits to what extent such conjugate properties maintain their approximate meaning, as the mathematical framework of quantum physics does not support the notion of simultaneously well-defined conjugate properties expressed by a single value. In quantum mechanics, the uncertainty principle (also known as Heisenberg's uncertainty principle) is any of a variety of mathematical inequalities asserting a fundamental limit to the accuracy with which the values for certain pairs of physical quantities of a particle, such as position, x, and momentum, p, can be predicted from initial conditions. Wikipedia Rate this definition: 0.0 / 0 votes 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
